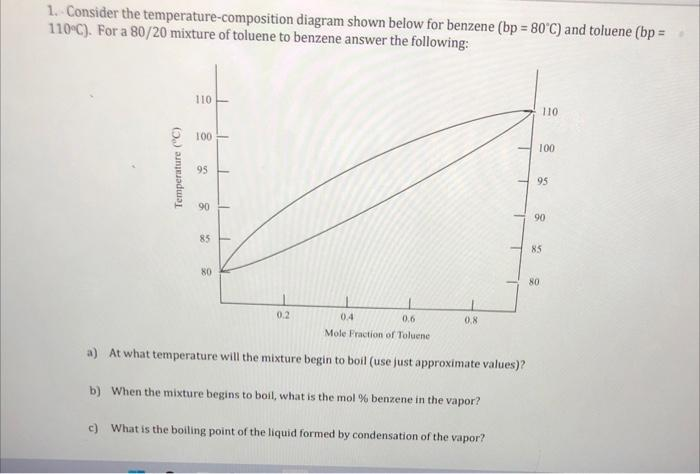
Consider the temperature-composition diagram shown below for benzene (bp = 80°C) and toluene (bp = 110°C). For a 80/20 mixture of toluene to benzene answer the following: Temperature (°C) 110 8 95 90 3 80 1 T 1 1 0.2 0.4 0.6 Mole Fraction of Toluene a) At what temperature will the mixture begin to boil (use just approximate values)? b) When the mixture begins to boil, what is the mol % benzene in the vapor? c) What is the boiling point of the liquid formed by condensation of the vapor? 1 0.8 110 100 95 90 85 80
The Correct Answer and Explanation is :
To analyze the problem, we will extract approximate values from the temperature-composition diagram provided. The key steps involve identifying the boiling temperature, calculating the vapor composition, and determining the boiling point of the condensed liquid.
Answers
a) At what temperature will the mixture begin to boil?
From the diagram, for a mixture with a mole fraction of 0.8 toluene (or 0.2 benzene), the boiling begins where the liquid composition intersects the liquidus curve (lower curve). For 80% toluene and 20% benzene, the approximate boiling point is about 96°C.
b) When the mixture begins to boil, what is the mol % benzene in the vapor?
At the boiling point, the vapor composition corresponds to the intersection of the vertical line (at 0.8 toluene mole fraction) with the vapor curve (upper curve). This point gives the composition of the vapor phase. From the diagram, the mole fraction of benzene in the vapor is approximately 0.55 or 55% benzene.
c) What is the boiling point of the liquid formed by condensation of the vapor?
The boiling point of the liquid formed by condensation corresponds to the liquid composition of the condensed vapor. The vapor initially at 55% benzene will condense to form a liquid at the liquidus curve. Tracing horizontally from the vapor composition (55% benzene) to the liquidus curve gives a boiling point of about 88°C.
Explanation (300 words)
The temperature-composition diagram illustrates phase behavior in binary mixtures. The liquidus curve represents the boiling points of liquid mixtures, and the vapor curve shows the composition of the vapor in equilibrium with the liquid at a given temperature.
For a mixture of 80% toluene (20% benzene), the boiling starts at the intersection of the liquidus curve with the 0.8 mole fraction toluene line. This is approximately 96°C. As the liquid boils, the composition of the vapor is richer in benzene due to its lower boiling point (80°C). From the vapor curve, this vapor is roughly 55% benzene.
When the vapor condenses, it forms a liquid with the same composition as the vapor. To find the boiling point of this new liquid, trace the vapor’s composition horizontally to the liquidus curve. At 55% benzene, the boiling point is around 88°C.
This process, known as fractional distillation, exploits the difference in boiling points to separate components of a mixture. Benzene’s lower boiling point makes it more volatile, so the vapor phase is enriched with benzene, whereas the liquid phase retains more toluene. This principle underpins industrial separation processes for hydrocarbons.